Present AI developments in drug improvement
Based on an {industry} survey, roughly 81% of pharma organizations now make the most of AI in not less than one improvement program1. Predictive modeling and new drug candidate identification and choice are among the many main workflows the place AI is being utilized in pharma R&D (Determine 1). This development is backed by staggering monetary commitments. Giant pharma gamers, similar to Pfizer, Takeda, and AstraZeneca, have ramped up their AI funding in recent times, with a selected concentrate on drug discovery.
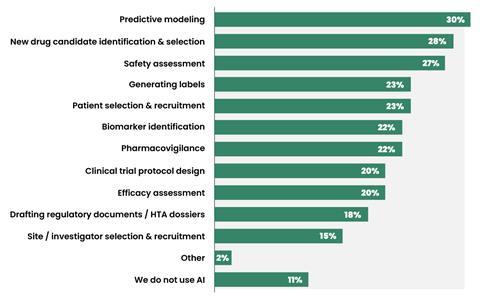
The worldwide AI-powered drug discovery market was valued at roughly $3.1 billion in 2025 and is predicted to rise to $4 billion in 20262. Prime AI-first biotechs embrace Isomorphic Labs (Alphabet), Insitro, Insilico Medication, and Recursion2. These key gamers are leveraging proprietary platforms and high-value collaborations with conventional pharmaceutical corporations to scale back improvement timelines, decrease general prices, and enhance the likelihood of drug candidate success. As an illustration, Isomorphic Labs signed AI drug discovery offers value practically $3 billion with pharmaceutical giants Eli Lilly and Novartis, and one other AI startup Chai Discovery introduced a partnership with Eli Lilly to speed up drug discovery utilizing generative design fashions3,4.
The function of high-throughput expression in AI design
As AI fashions change into more and more adept at de novo design, the demand for fast moist lab validation has skyrocketed5,6. Sino Organic’s high-throughput (HTP) antibody manufacturing platforms primarily based on twin expression methods–mammalian and cell-free methods–have emerged as transformative instruments able to quickly producing hundreds of AI-designed antibody variants. This permits researchers to maneuver seamlessly from AI-generated sequences to purposeful antibodies in simply days, supporting the quick design-build-test-learn cycles required to refine AI fashions (Determine 2).
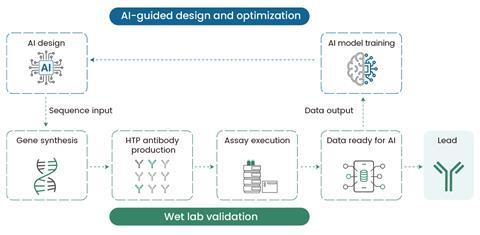
Confirmed mammalian excellence & fast cell-free innovation
Conventional mammalian expression methods, similar to HEK293 and CHO cell strains, stay the {industry} gold commonplace for HTP manufacturing of recombinant antibodies, together with full-length IgG, VHH, and scFv. Sino Organic leverages deep experience in HTP gene synthesis, vector development, and optimized transient antibody expression expertise to provide small-scale, high-quality recombinant antibodies, delivering industry-leading throughput (10,000+ antibodies/month) and velocity (10 days from gene to antibody). Moreover, instant entry to a catalog of 10,000+ premium recombinant proteins permits for fast, correct binding validation of those antibodies, making certain specificity and high-quality information.
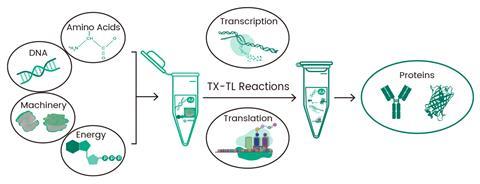
Determine 3. Cell-free protein synthesis course of.
Case research of HTP scFv-His and VHH-His synthesis through CFPS: The HTP functionality of Sino Organic’s CFPS platform was lately demonstrated in a mission involving over 2,000 AI-designed scFv and VHH sequences (Determine 4). Your complete library was synthesized and expressed in parallel, adopted by binding affinity evaluation through BLI. This built-in HTP workflow–bridging manufacturing and characterization–facilitated the environment friendly identification of promising leads with picomolar affinity and the technology of important information for additional pipeline optimization.
Format: 2,000+ scFv-His and VHH-His antibodies
Goal: Completed expression and affinity evaluation with totally different goal proteins by BLI u464
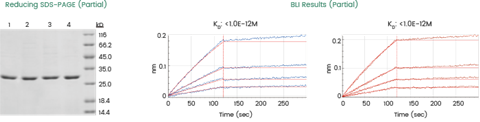
u464
Early-stage developability profiling is pivotal for de-risking the drug discovery pipeline and streamlining candidate choice8,9. Complete antibody developability analysis permits the proactive identification and administration of potential roadblocks, similar to filtering out candidates with appropriate binding traits however undesirable biophysical properties.
Desk 1. Antibody developability assays at Sino Organic.
| Class | Assays |
|---|---|
| Purity | SEC-HPLC/SDS-PAGE |
| Titer | ELISA |
| Solubility | DLS |
| Intact Mass | LC-MS |
| Colloidal Stability | SMAC-HPLC |
| Thermo Stability | nanoDSF/DSC |
| Aggregation | SEC-HPLC/SEC-MALS |
| Self-Affiliation | AC-SINS |
| Hydrophobicity | HIC-HPLC/PAIA-HIC |
| Dimension Distribution and Aggregation | DLS |
| Goal Binding | ELISA/SPR/BLI |
| Affnity Evaluation | FACS/SPR/BLI |
| FcγR/FcRn/C1q Binding | SPR/BLI |
| Polyreactivity | BVP/DNA/Insulin ELISA |
| Cell-Primarily based Assay | Assay Particular |
Synchronizing discovery: An Built-in HTP platform for AI-driven innovation
As AI-driven innovation accelerates, aligning experimental throughput with the size of recent discovery is paramount. That is particularly vital as antibody pipelines increase, fueling the demand for seamless HTP expression and parallelized assay workflows.
References
1. Norstella. Assessing the affect of AI transformation on pharma R&D. [Online]. Accessible at: https://www.norstella.com/assessing-ai-transformation-pharma-rd/ (Accessed: 24 March 2026).
3. Isomorphic Labs. Isomorphic Labs kicks off 2024 with two pharmaceutical collaborations. [Online]. Accessible at: https://www.isomorphiclabs.com/articles/isomorphic-labs-kicks-off-2024-with-two-pharmaceutical-collaborations (Accessed: 24 March 2026).
4. TechCrunch. From OpenAI’s workplaces to a cope with Eli Lilly: How Chai Discovery turned one of many flashiest names in AI drug improvement. [Online]. Accessible at: https://techcrunch.com/2026/01/16/from-openais-offices-to-a-deal-with-eli-lilly-how-chai-discovery-became-one-of-the-flashiest-names-in-ai-drug-development/ (Accessed: 24 March 2026).
6. Li X, et al. Superior drug supply methods and AI-driven formulation optimization. Pharmaceutics. 2023; 15(7): 1916. https://doi.org/10.3390/pharmaceutics15071916
7. Wang Y, et al. Monoclonal antibody developability evaluation: Challenges and alternatives. Antibodies. 2015; 4(1): 12. https://doi.org/10.3390/antib4010012
9. Nature Biomedical Engineering. Navigating the interface of AI and moist lab automation. Nat Biomed Eng. 2025; 9: 00349-8. https://doi.org/10.1038/s44222-025-00349-8


